Learn More: High-Capacity Nitrogen Generators
High-Capacity Nitrogen (N2) Applications
Nitrogen's inert properties make it a necessity for many industries. It's widely used for drying, purging, cooling, and to create protective barriers between sensitive parts and the damaging environment.
In pharmaceutical or life science laboratories, N2 works within desiccators and glove boxes to protect moisture-sensitive parts and samples. Other industries that rely on the drying properties of N2 are semi-conductor and electronics.
Food packaging lines are a good example of where high volumes of nitrogen are used to protect consumables from spoiling by contact with oxygen. The N2 creates a protective barrier. The same idea can be applied to flammable raw materials: contact with oxygen would be harmful.
Nitrogen is incorporated into heat-treating, welding and laser-cutting processes in the steel and aerospace industries. For chemicals production, pressurized N2 can propel liquids through pipelines and help remove volatile organic chemicals (VOCs) from manufacturing environments.
Nitrogen is the most abundant gas present in the air we breathe. By separating it into a pure gas, many useful applications have been developed. Generating large quantities of N2 as needed is a convenient, cost-effective practice.
N2 Generator System Components:
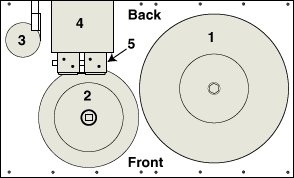
- 1. 200-gallon compressed air holding tank
- 2. 60-gallon nitrogen holding tank
- 3. Hydrocarbon filter
- 4. Two (2) nitrogen membranes and one (1) exit filter
- 5. Coalescing pre-filters
- 6. High-volume rotary screw compressor
Specifications
- Nitrogen Purity: 99%+ (depending on inlet pressure)
- Power Requirements (compressor): 208/230/460 VAC, 60Hz, 3-phase, prewired for 460V operation
- Power Requirements (generator): 120VAC / 240VAC
- Generator Max. Power: 4kW
- Footprint: 60"W x 44"D (generator); 90W" x 72"D (generator and compressor)
- Maximum height: 96"
- Generator Tank Capacity N2 holding tank: 60 gal.; Compressed air storage: 200 gal.
- Compressor* pressure range: 217 psig
- Compressor* Capacity: 157 ACFM
- Compressor* Motor Power: 50 HP
- Compressor* Dimensions: 78.375"L x 40.5"W x 67"H
- Compressor Weight: 2,458 lb
High-Capacity Nitrogen Membrane Performance
| Test # | Inlet Pressure (psi) | Pressure Entering N2 Membrane (psi) | Pressure in N2 Holding Tank (psi) | Outlet Flow Rate (cfm) | % N2 | Comments |
|---|---|---|---|---|---|---|
| 1 | 120 | 120 | 25 | 13 | 99 | 0% RH |
| 2 | 120 | 120 | 35 | 18 | 99 | 0% RH |
| 3 | 120 | 120 | 80 | 30 | 99 | 0% RH |
| 4 | 120 | 100 | 60 | 18 | 99 | 0% RH |
| 5 | 120 | 100 | 50 | 15 | 99 | 0% RH |
| 6 | 120 | 90 | 50 | 20 | 99 | 0% RH |
| 7 | 120 | 90 | 40 | 13 | 99 | 0% RH |
